
Bacterial estrogenesis without oxygen: Wood-Ljungdahl pathway likely contributed to the emergence of estrogens in the biosphere
Androgen and estrogen, key sex hormones, were long thought to be exclusively produced by vertebrates. The O2-dependent aromatase that converts androgen to estrogen (estrogenesis) has never been identified in any prokaryotes. Here, we report the discovery of anaerobic estrogenesis in a Peptococcaceae bacterium (Phosphitispora sp. strain TUW77) isolated from the gut of the great blue-spotted mudskipper (Boleophthalmus pectinirostris). This strain exhibits unprecedented testosterone fermentation pathways, transforming testosterone into estrogens and androstanediol under anaerobic conditions. Physiological experiments revealed that strain TUW77 grows exclusively on testosterone, utilizing the androgenic C-19 methyl group as both the carbon source and electron donor.
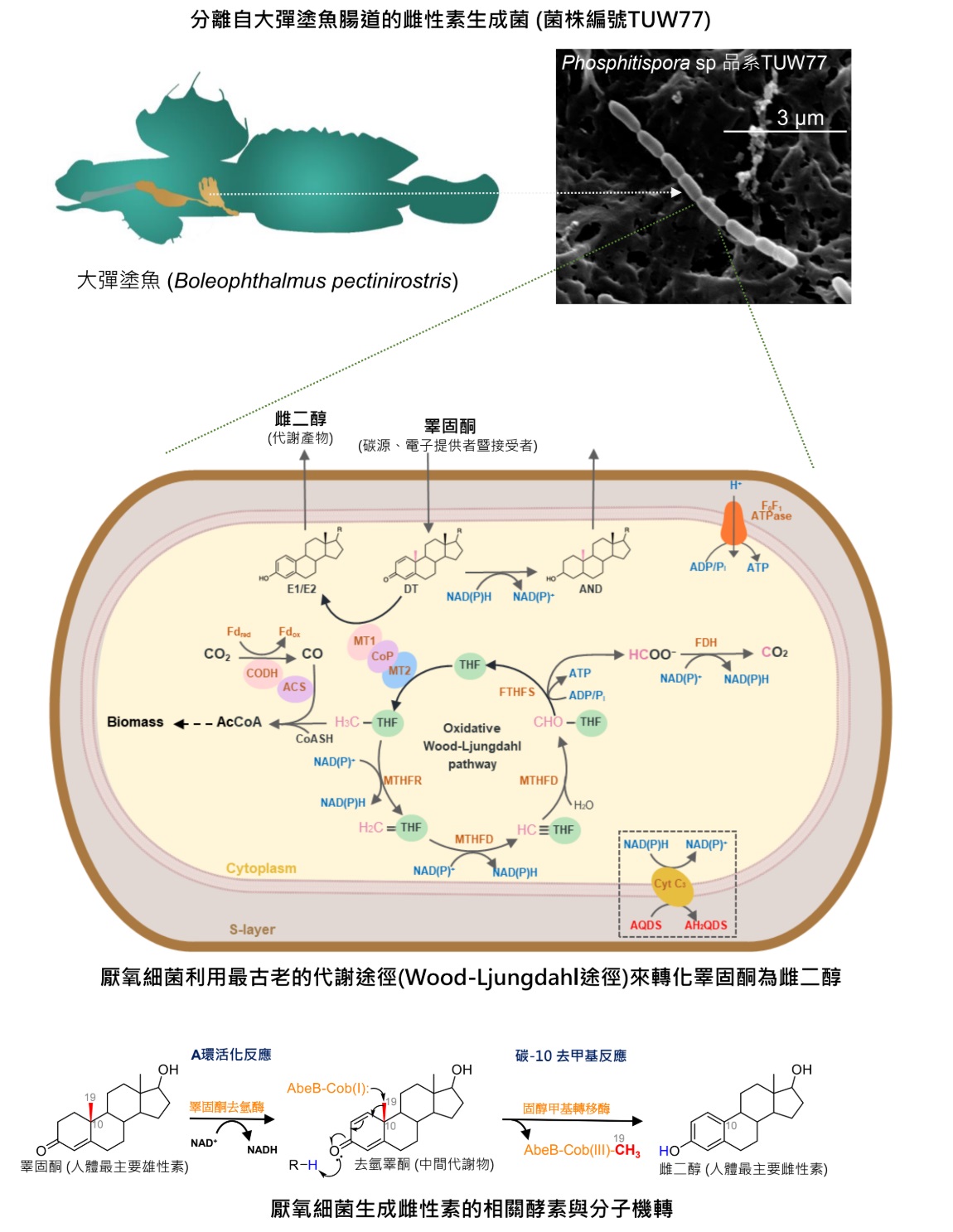
The genomic analysis identified three copies of a polycistronic gene cluster, abeABC (anaerobic bacterial estrogenesis), encoding components of a classic cobalamin-dependent methyltransferase system. These genes, highly expressed under testosterone-fed conditions, show up to 57% protein identity to the characterized EmtAB from denitrifying Denitratisoma spp., known for methylating estrogen into androgen (the reverse reaction). Tiered transcriptomic and proteomic analyses suggest that the removed C-19 methyl group is completely oxidized to CO2 via the oxidative Wood-Ljungdahl pathway, while the reducing equivalents (NADH) fully reduce remaining testosterone to androstanediol. Consistently, the addition of anthraquinone-2,6-disulfonate, an extracellular electron acceptor, to testosterone-fed TUW77 cultures enabled complete testosterone conversion into estrogen without androstanediol accumulation (anaerobic testosterone oxidation).
This discovery of aromatase-independent estrogenesis in anaerobic bacteria suggests that the ancient Wood-Ljungdahl pathway may have contributed to the emergence of estrogens in the early biosphere. Our findings challenge the long-held belief that estrogen production is exclusive to aromatase-containing vertebrates, expanding our understanding of steroid hormone biosynthesis across domains of life. The involvement of ancient strictly anaerobic Peptococcaceae members and the Wood-Ljungdahl pathway suggests that bacterial estrogenesis may predate O2-dependent estrogenesis in vertebrates. As estrogens are potent endocrine disruptors, the discovery of bacterial estrogenesis reveals a previously unknown source of environmental estrogens. Furthermore, the identification of estrogen-producing bacteria in animal guts opens new avenues for potential microbiome-based hypoestrogenism therapies to supplement estrogen in menopausal or ovariectomized females, offering an innovative alternative to current hormone replacement strategies. The research has been published on March 8, 2025 in PNAS.
Article link: https://www.pnas.org/doi/10.1073/pnas.2422930122
Related link: https://biodiv.tw/research-20241111190746





